Resources
Downloadable materials
Crysvita® Resources
XLH Resources
Symposium Highlights
Videos
Publications of interest
Key facts
Key Facts about XLH and CRYSVITA®
XLH
What is XLH?
XLH is a rare, hereditary, progressive and lifelong renal phosphate wasting disorder caused by mutations in the PHEX (phosphate-regulating endopeptidase homolog, X-linked) gene that leads to excess activity of fibroblast growth factor 23 (FGF23)1–4
What is the prevalence of XLH?
XLH is a rare disease that affects approximately 1 in 20,000–60,000 people1,5

How is XLH inherited?
XLH is inherited in an X-linked dominant pattern; however, 20–30% of cases arise from spontaneous mutations6,7
What is XLH caused by?
XLH is caused by mutations in the PHEX gene4,5, which is located on the X chromosome
What does it mean for patients with XLH?
Excess FGF23:
- Decreases renal phosphate reabsorption, which increases urinary phosphate excretion8
- Decreases active vitamin D (1,25[OH]2D) production, which reduces intestinal phosphate absorption8
The resulting chronic hypophosphataemia impairs bone mineralisation, leading to a variety of clinical manifestations that can impair patients’ physical function and quality of life9
XLH is not just a bone disease – it is a multisystemic disease that impacts muscles and dentition as well4,10
CRYSVITA®
What is CRYSVITA® ?
CRYSVITA® is a recombinant, fully human monoclonal antibody IgG1 that binds to and inhibits excess FGF23 activity.11 It is the first and only disease-modifying biologic treatment that targets the pathophysiology of XLH.12
How does CRYSVITA® work?
By inhibiting excess FGF23 activity, CRYSVITA® helps restore phosphate homeostasis in people with XLH and improves bone mineralisation mobility and pain.11-14
Who can receive CRYSVITA®?
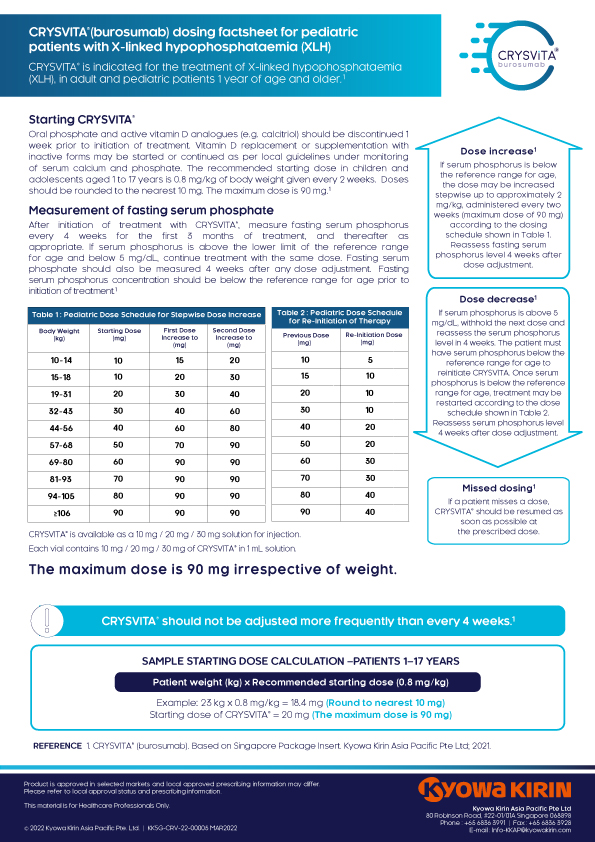
CRYSVITA® is indicated for the treatment of X-linked hypophosphatemia (XLH) in adult and pediatric patients 1 year of age and older.11
Why use CRYSVITA®?
The efficacy and safety of CRYSVITA® in children aged 1–12 years, and adults, with XLH have been studied in a global clinical development programme.12–16
A phase 3 clinical study in children with XLH showed that compared with continuing conventional therapy, switching children to CRYSVITA®13:
- Improved phosphate homeostasis
- Significantly improved rickets healing and reduced its severity up to Week 64
- Significantly improved growth and mobility outcomes up to Week 64
- Significantly improved biochemical markers of phosphate regulation and bone health up to Week 64
- In this phase 3 clinical study, CRYSVITA had an acceptable safety profile over 64 weeks in children with XLH13
Phase 3 clinical studies in adults with XLH:
- Phosphate homeostasis, fracture healing, bone mineralisation and remodelling improved, and stiffness were reduced in the CRYSVITA® group compared with the placebo group in a double-blind placebo-controlled study.16
- Phosphate homeostasis improved, and bone quality, mineralisation and remodelling increased in patients treated with CRYSVITA® by Week 48 when compared with that at baseline in a single-arm study.14
- There was more healing of baseline fractures/pseudofractures in patients who continued CRYSVITA® compared with those who received CRYSVITA® after placebo at Week 48 in an open-label study.12
- When placebo-treated patients started CRYSVITA® treatment at Week 24, the healing of fractures/pseudofractures at Week 48 was similar to the healing at Week 24 in those who received CRYSVITA® therapy from the beginning of the study.12
- CRYSVITA® led to sustained improvements in pain, stiffness and physical function and mobility at Week 48 when compared with that at baseline in a double-blind placebo-controlled study.12
- In these phase 3 studies, CRYSVITA® had an acceptable safety profile up to 48 weeks in adults with XLH.12,14
Useful Websites
Publicly Available Websites
XLH information websites for healthcare professionals

1. XLHLink.asia (Healthcare professional website)
https://www.xlhlink.asia/hcp/
This website is developed and funded by Kyowa Kirin Asia Pacific Pte. Ltd.
XLH information websites for healthcare patients

1. XLHLink.asia (Patient website)
https://www.xlhlink.asia/
This website is developed and funded by Kyowa Kirin Asia Pacific Pte. Ltd.

2. Shine A Light on XLH
https://shinealightonxlh.com/tc/
This website is developed and funded by Kyowa Kirin Asia Pacific Pte. Ltd
1. Beck-Nielsen SS, et al. Eur J Endocrinol. 2009;160:491–7. 2. Endo I, et al. Endocr J. 2015;62:811–6. 3. Carpenter TO, et al. J Bone Miner Res. 2011;26:1381–8. 4. Haffner D, et al. Nat Rev Nephrol. 2019;15:435–55. 5. Rafaelsen S, et al. Eur J Endocrinol. 2016;174:125–36. 6. Rajah J, et al. Eur J Pediatr. 2011;170:1089–96. 7. Raimann A, et al. Wien Med Wochenschr. 2020;170:116–23. 8. Razzaque MS. Nat Rev Endocrinol. 2009;5:611–9. 9. Linglart A, et al. Endocr Connect. 2014;3:R13–30. 10. Beck-Nielsen SS, et al. Orphanet J Rare Dis. 2019;14:58. 11. CRYSVITA® (burosumab). Based on Singapore Package Insert. Kyowa Kirin Asia Pacific Pte Ltd; May 2021. 12. Portale AA, et al. Calcif Tissue Int. 2019;105:271–84. 13. Imel EA, et al. Lancet. 2019;393:2416–27. 14. Insogna KL, et al. J Bone Miner Res. 2019;34:2183–91. 15. Carpenter TO, et al. N Eng J Med. 2018;378:1987–98. 16. Insogna KL, et al. J Bone Miner Res. 2018;33:1383–93